Increase Profit by Substantially Increasing Speed-to-Market
While reducing risk of audits and product recalls,with our Enterprise Quality Management Software System
1000+ Companies use our EQMS
Used by 5 of the Largest Regulatory Agencies in the World
Used by 3 of the Top 5 Life Science Companies
-
#1 in EQMS since 1993
-
1st to be 21CFR Part 11 compliant
-
100% built by industry experts
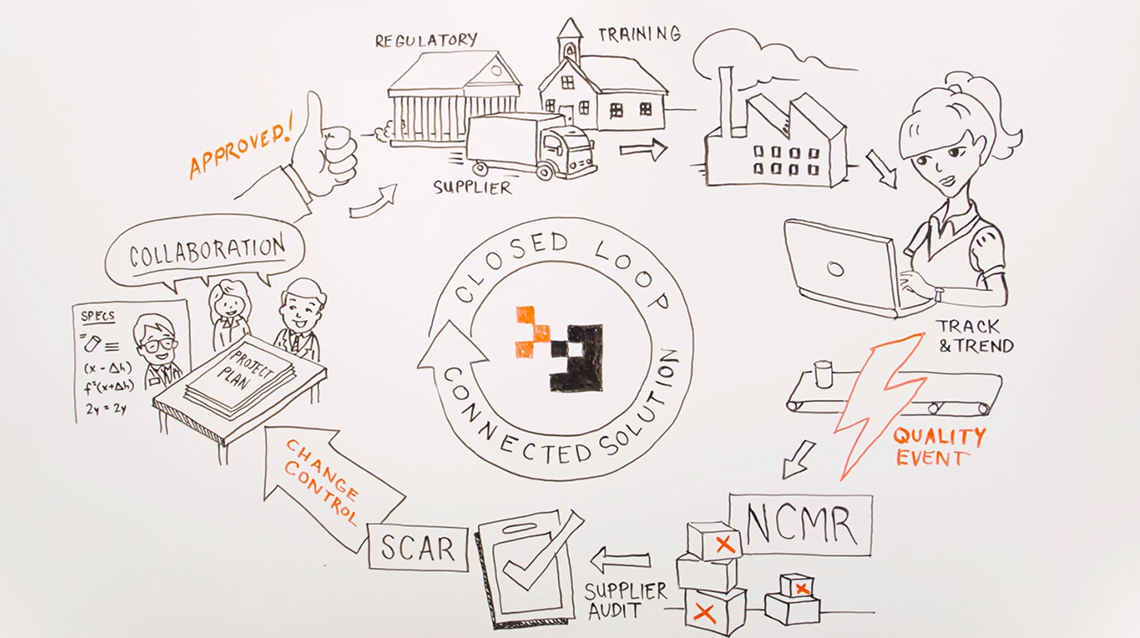
MasterControl's integrated quality management solutions facilitate compliance with regulations and standards such as:
-
- 21 CFR Part 11
- 21 CFR Parts 210 – 211
- 21 CFR Part 820
- 21 CFR Part 606
-
- ISO 9000 Standards
- ISO 14000
- ISO 13485
- TS 16949
-
- CLIA Regulations
- Canadian Quality Standards
- European Union Standards
Understanding your business needs and offering a solution that meets your needs is essential. A PBS consultant will work closely with your team to assess your QMS needs in order to define a suitable and cost effective solution.
A PBS consultant and, if necessary, MasterControl consultants can also provide process consulting. This service focuses on investigating requirements for a GxP or business process and then creating a design based on best practices, which includes the form(s) and other elements necessary to enable the defined process. The deliverable of this service is a design document that provides clear specifications necessary for the successful implementation of a process by either the customer or a MasterControl service professional. This may require on-site service for requirements gathering.
Project Stage:
PBS will nominate a project manager who will lead the project team assigned to your project. He or she will be responsible for implementing the project from the initial kick-off meeting all the way to going live. For customers requiring validation, their services include validating the system to meet the regulatory requirement. PBS project implementation is based on MasterControl’s successfully proven methodology and ensures high quality.
After-Sales Stage:
Besides the standard warranty services, PBS can provide after sales-support. PBS technical support teams are available 9 a.m. to 5 p.m. weekdays to answer your questions.
As your need for QMS grows or as you decide to incorporate more of MasterControl’s available functionality, the PBS team will be available to support your expansion of the system.
PBS engineers can also provide assistance in the maintenance of your system and help keep the system in validated state as part of a service contract.